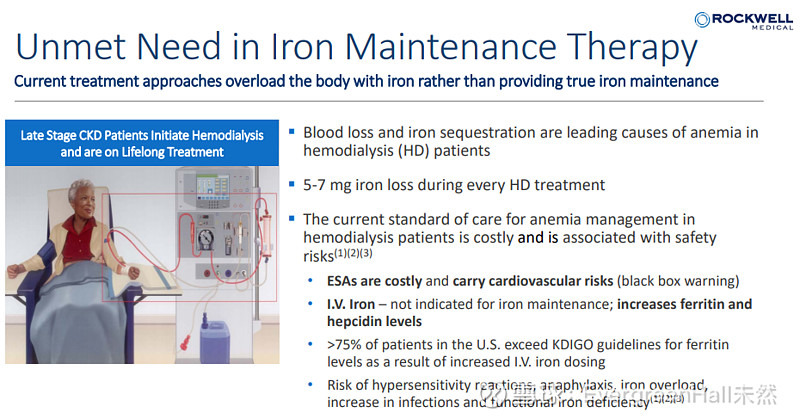
$罗克韦尔医药(RMTI)$ 本次PDUFA是递交静脉注射版本Triferic,与血液透析版的Triferic进行生物等效性研究,已经获批。
这种NDA的方式之前获得FDA同意
静脉注射的剂型有什么好处: An I.V. formulation would give us the opportunity to integrate the use of Triferic at all dialysis centers— in the U.S. and abroad—regardless of the mode of bicarbonate delivery being used.,大概的意思是使用场景限制更低。
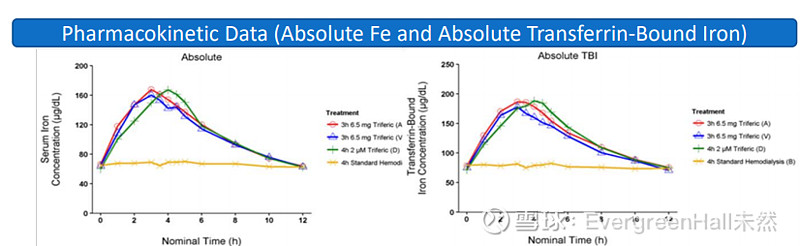
研究结果显示注版月血液透析版输送的铁量相同
对24例CKD-5HD患者进行了开放性、随机、多期单剂量研究,以确定透析液和静脉注射之间的剂量等效性。每位患者接受一个基线铁治疗和三个随机治疗,分别是三联铁6.75 M g静脉注射预透析器;三联铁6.75 mg静脉注射后透析器;或三联铁2μM(110μg Fe/L血液透析液)。在指定时间点超过12小时抽取血清和血浆铁,分析总铁(Fe)和转铁蛋白结合铁(TBI)。研究结果显示:在血液透析的3小时内静脉输注6.5毫克铁作为柠檬酸焦磷酸铁(FPC),所输送的铁量与通过血液透析版相同。在既往的3期临床研究中,透析液三价铁维持铁状态和血红蛋白浓度;在透析液三价铁的关键临床试验中输送的铁量现在确定为6.5毫克铁/治疗;以及将FPC注入预透析器的血液中,可以为所有接受血液透析或血液透析滤过的患者提供三价铁。
附2019 ADC会议的摘要
Ferric Pyrophosphate Citrate (Triferic) Administered Via Dialysate Provides 6.5 Mg of Iron at Each Treatment.
Raymond Pratt, Ajay Gupta. 1Rockwell Medical Inc., Wixom, Michigan, USA
Introduction and Aims: Ferric pyrophosphate citrate (FPC, Triferic®) is approved in U.S.A as an iron replacement product to maintain hemoglobin when added to the bicarbonate concentrate and administered through the dialysate A new FPC presentation containing 6.75 mg iron in 4.5 mL was developed for intravenous (i.v.) administration during hemodialysis. The aim of this study was to establish the equivalence between iron delivery via dialysate and i.v. infusion using a pharmacokinetic approach.,
Methods:An open-label, randomized, multiple period single dose study was conducted in 24 CKD-5HD patients to establish the equivalence of doses between dialysate and i.v administration. Each patient received a baseline and 3 randomized treatments. Serum and plasma iron was drawn at specified time points over 12 hours and analyzed for total Fe and Transferrin Bound Iron (TBI). The treatments were 1) basal iron profile over 12 hours; 2) Triferic 6.75 mg Fe IV pre-dialyzer (Pre-D); 3) Triferic 6.75 mg Fe IV post-dialyzer (Post-D) and 4) Triferic 2 μM (110 μg Fe/L of hemodialysate (Fe-HD). Bioavailability was determined by comparing Cmax and AUC0-t of Fe-HD (reference) and test treatments Fe Pre-D (Arterial) and Post-D (venous) iron profiles.,
Results: The serum iron vs. time profile was similar between HD and IV administered FPC for absolute plasma Fe as well as TBI. Calculated bioavailability parameters showed that infusion of 6.5 mg of FPC iron i.v. delivered equivalent amounts of iron as 4 hours via hemodialysate (110 μg Fe/L).
Conclusions: Intravenous infusion of 6.5 mg iron as ferric pyrophosphate citrate (FPC, Triferic®) during 3 hours of HD delivers the same quantity of iron as when Triferic is delivered via hemodialysate. The Phase III clinical studies of FPC maintained iron status and hemoglobin concentrations when delivered via dialysate regularly. The amount of iron delivered in the pivotal clinical trials is now established as 6.5 mg Fe/treatment. Administration of FPC into the pre-dialyzer blood line extends the ability to provide Triferic iron to all patients receiving hemodialysis or hemodiafiltration.