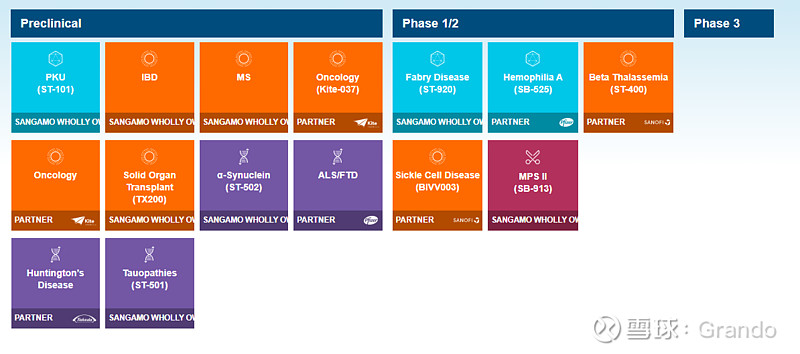
$Sangamo Therapeutics(SGMO)$ SB-318 怎么不在pipeline里面了?没看到有公告啊。
| 发布于: | 雪球 | 转发:0 | 回复:1 | 喜欢:0 |
$Sangamo Therapeutics(SGMO)$ SB-318 怎么不在pipeline里面了?没看到有公告啊。

We have three proprietary in vivo genome editing programs being evaluated in Phase 1/2 clinical trials: SB-913 (Mucopolysaccharidosis type II, or MPS II), SB-318 (MPS I), and SB-FIX (hemophilia B). In April 2019, we announced that we are no longer treating additional patients in our SB-913, SB-318 and SB-FIX clinical trials with first generation ZFNs given that clinical benefit has not been demonstrated in analyses conducted to date.
We are planning a new clinical trial for SB-913 to treat MPS II to evaluate second-generation ZFNs and other potential modifications that have the potential to enhance the clinical efficacy of this product. These include modifications that have the potential to enhance the in vivo delivery of the ZFNs.